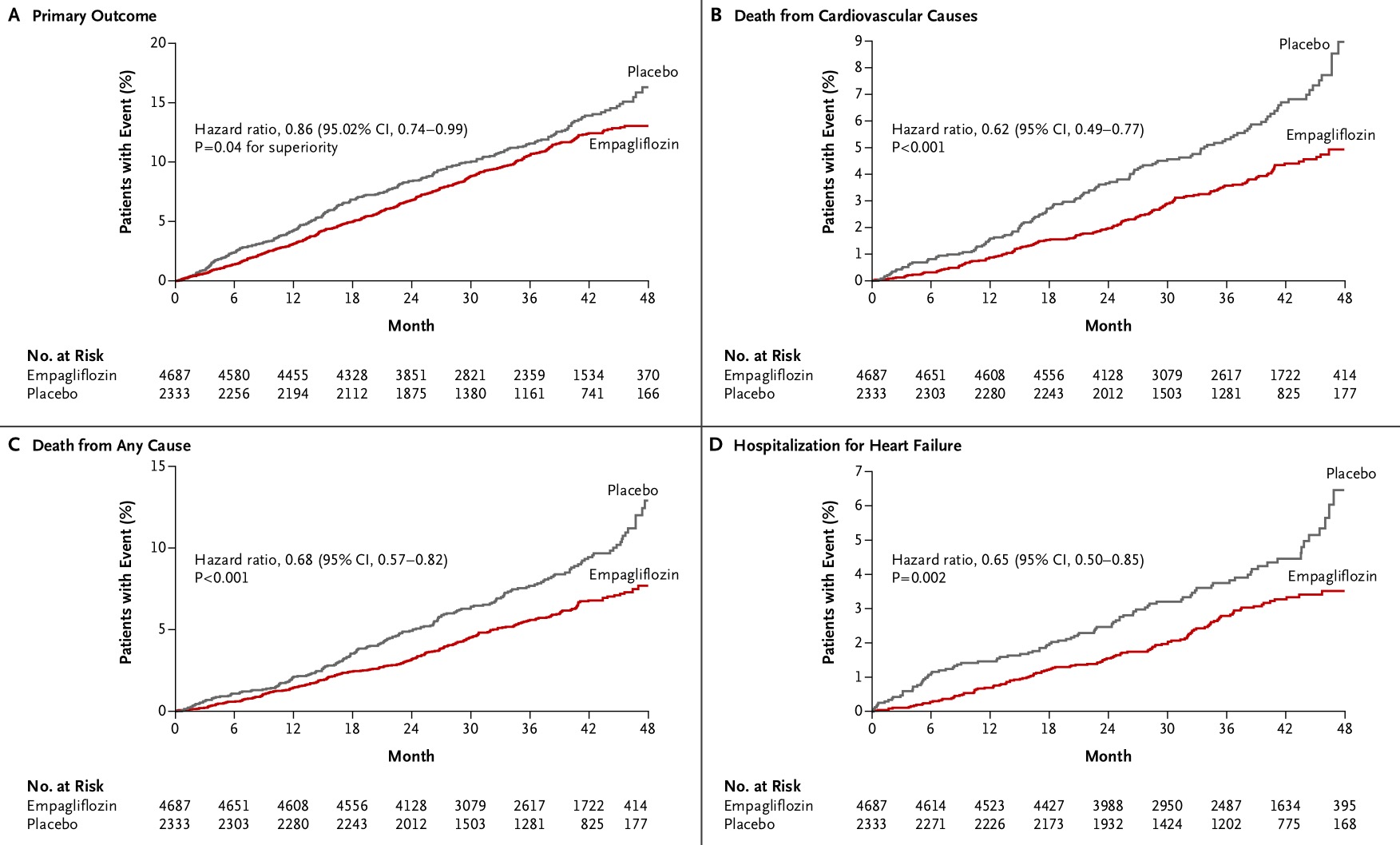
Probably for the first time a glucose-lowering drug has shown significant 38% relative reduction in cardiovascular mortality and a 32% relative reduction all cause mortality. Empagliflozin, a sodium-glucose cotransporter 2 (SGLT2) inhibitor reduces serum glucose levels by cutting down renal reabsorption and increasing urinary excretion of glucose. A large industry sponsored randomized trial examined the effects of empagliflozin on cardiovascular outcomes in 7020 type 2 diabetes patients (mean age 63 years) with previous history of coronary artery disease, stroke, or peripheral arterial disease (EMPA-REG OUTCOME Trial).
All patients at baseline had glycosylated hemoglobin (HbA1C) levels between 7% and 10%. Most patients had long-standing diabetes and were on anti-diabetic, anti-hypertensive and statin medication. Patients were randomized to 10 mg of empagliflozin, 25 mg of empagliflozin or placebo for around 3 years.
The reduction in the composite of clinical outcome was driven largely by the reduced cardiovascular deaths. The outcomes in the two empagliflozin groups were similar and hence they were pooled. The primary composite outcome (CV-related death, nonfatal myocardial infarction, or nonfatal stroke) occurred significantly less in the treated group (10.5%) than with placebo (12.1%), p = 0.04. There was a 2.2% reduction in cardiovascular death (3.7% vs. 5.9%; p< 0.001). The reduction in mortality was however not accompanied by significantly reduced myocardial infarction or stroke.
Recently it was shown that lixisenatide (GLP-1; glucagon like receptor analogue) and saxaglaptin (DPP 4 inhibitor) neither increased nor decreased cardiovascular risk in patients with diabetes. It is for the first time that an anti-diabetic drug (empagliflozin) has demonstrated clear cardiovascular superiority while treating diabetic patients. Diabetes is a dreaded risk factor for coronary disease and stroke but ironically for all these years the treating physician did not have a single drug to lower this risk.
The investigators have so far been unable to explain reduced mortality without any anti-atherosclerotic effect. The life expectancy of diabetic patients at high cardiovascular risk is reduced by almost 12 years on an average. Empagliflozin was associated with fewer deaths after myocardial infarction, fewer deaths in heart failure patients and less deaths of unknown cause. There was increased rate of genital infections with empagliflozin compared with placebo (6.4% vs. 1%, p < 0.01).
There was also a significant 35% relative reduction in heart failure hospitalization with empagliflozin and this may explain reduction in mortality without reduction in myocardial infarction or stroke. Empagliflozin, by promoting osmotic diuresis, may be correcting diastolic heart failure or early systolic heart failure and preventing sudden cardiac death due to arrhythmias. It is unclear at this stage whether the cardiovascular benefits of empagliflozin can be extrapolated to other SGLT2 inhibitors. The cost of this new drug is expected to be a stiff US $ 5,000 annually and therefore for every 50 patients treated for 3 years to benefit 1 person the economic burden will be almost $750,000 for that duration.
The mechanisms behind the cardiovascular benefits of empagliflozin besides diuresis could be improvement in arterial stiffness, lesser cardiac oxygen demand, reduction in albuminuria, lower serum uric acid level, lesser visceral fat and improvement in blood pressure. Interestingly empagliflozin was significantly effective when HBA1C was less than 8.5% and in those with body mass index (BMI) equal or more than 30 and not in those more than 30. The EMPG-REG Outcome Trial has shown for the first time that a glucose lowering drug given to patients with type 2 diabetes and established cardiovascular disease may lower rate of death and hospitalisation for congestive heart failure.